
by Petr Gebousky, Miroslav Kárny and Hana Krizová
Lymphoscintigraphy is an emerging technique for judging of the state of lymphatic system. It serves well for diagnosis of lymphedema. Its early and correct detection prevents a range of complications leading often to a full disability. A wider use of this technique is inhibited by a lack of a reliable quantitative evaluation of its results especially in difficult case of upper limbs in early stage of the disease. The modeling and subsequent identification can be exploited in order to improve the accuracy of diagnosis.
Lymphedema is a progressive condition characterized by four pathologic features: excess tissue protein, edema, chronic inflammation and fibrosis. Upper limb lymphedema is a frequent complication of breast cancer.
A variety of imaging modalities have been employed in the evaluation of the lymphatic system. Lymphoscintigraphy is a noninvasive technique in which radionuclides are used to image regional lymph drainage systems.
It provides information about lymph transportation, filtration and reticuloendothelial function in extremities.
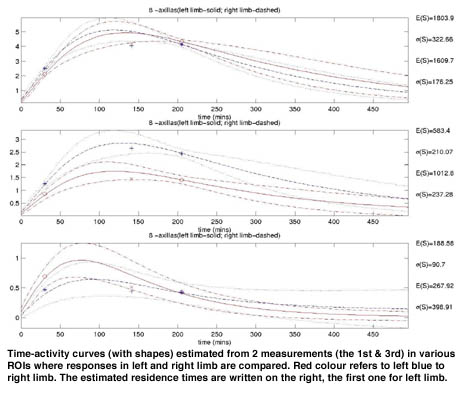
Standard inspection method consists of administration of 25 MBq of Tc-99m labeled colloid administered to the 1st interstitial space of both hands. Static 1 min images are taken immediately, 30 & 180 min after administration, morphologically evaluated and arm results semi-quantitatively compared. No standard is available for healthy population.

The system identification is employed here for description of individual response of the patient. We tried to model the accumulation of the radioactive tracer in local regions of upper limb during lymphoscintigraphic procedure by a chain of simple identical compartmental models with a common time constant, unknown gain and unknown number of constituents. The chosen 3-parameters model was motivated by a standard modeling of distributed parameter systems. Their Bayesian estimator was designed that converts integral counts taken over regions of interests (ROI : forearm, upper arm, axilla) into posterior distribution of discretised common time constant, model order and gain. Each triple of parameters determines uniquely time-activity curve in individual ROIs together with the important parameter for diagnosis - area under curve (residence time). This allows us to compute posterior distribution of residence time in each ROI.
Having in these distributions in hands, we can (i) judge (in)significance of differences between both arms (ii) study quantitative relationships between disease staging and measurements (iii) optimize measurements moments.
The proposed model and data processing were tested on data of 24 patients that were measured more often than usual (up to 5 measurements). Real data was provided by Department of Nuclear Medicine FNM. The evaluation results indicate adequacy of the adopted model and encourage us to follow the direction outlined under (i) to (iii).
Please contact:
Petr Gebousky - CRCIM (UTIA)
Tel: +420 2 66052583
E-mail: gebousky@utia.cas.cz